Clinical trials

We provide secure, compliant logistics for clinical trials, keeping products safe, traceable, and on schedule. With our global network, we can fulfil even the most specific requests from clinical trial partners.
What we do
Driving smarter access and sustainable margins
We manage the logistical flow of products used in veterinary clinical trials, monitoring every step closely. Our role begins with planning and documentation, followed by temperature-controlled transport and precise tracking from dispatch to delivery.
By maintaining product integrity and full traceability, we help research partners conduct their studies safely, efficiently, and in accordance with international requirements.
Active in more than 50 countries
Our team of experts
Global expertise.
Local commitment.
We cover it all
We handle complexity – you stay in control.
01
Logistics & Delivery
We manage logistics and delivery down to the finest detail. From storage to final transport, every step is carefully planned and executed to ensure products arrive efficiently, reliably, and on time.
02
Compliance
We operate under strict pharmaceutical guidelines, making sure that every process lives up to our standards.
03
Documention
Every shipment includes the right supply information to ensure transparency and compliance. Documentation is not just paperwork, but an essential part of quality assurance.
04
Regulatory Expertise
Our team is trained in GDP regulation, which allows us to provide service at the highest professional level.
Animals
Access en margin optimalisation for all animals
Whether pets, livestock, or specialty species — Zovea Animal Health provides the right products and services to keep animals healthy and care running smoothly.
Our approach
Controlled logistics for research materials
We coordinate the movement of investigational veterinary products within controlled and compliant frameworks. Each shipment is prepared according to study protocols and regulatory requirements, with validated temperature control and continuous monitoring to protect product integrity.
We manage documentation, labelling, and chain-of-custody records to ensure full traceability from manufacturer to study site. This structured process allows research partners to focus on their studies while maintaining confidence in the safety and reliability of each delivery.
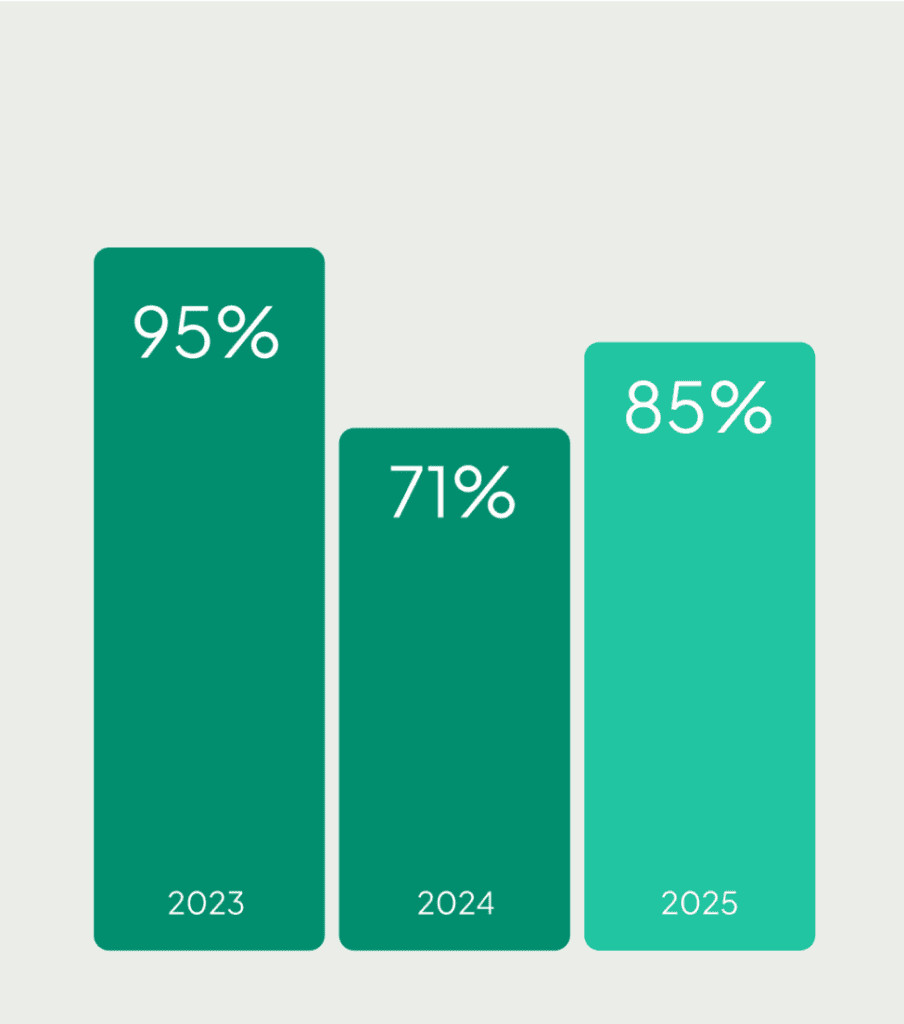
Veterinary Clinical Trials becomes smarter, faster, and safer.
Veterinary Clinical Trials becomes smarter, faster, and safer.
FAQ
Share your challenge, request your solution.
Tell us about your Clinical Trial needs, and we will create a tailored solution to keep your research moving.

Commercial Manager
Timo Lugard














